Sample is titrated from its initial pH to four pH points located close to 6.7, 5.9, 5.2 and 4.3 on the pH scale. Volatile fatty acid (VFA) and alkalinity concentrations are calculated with a personal computer program - because the titration method uses 5 pH points it is called 'Five pH point titration'.
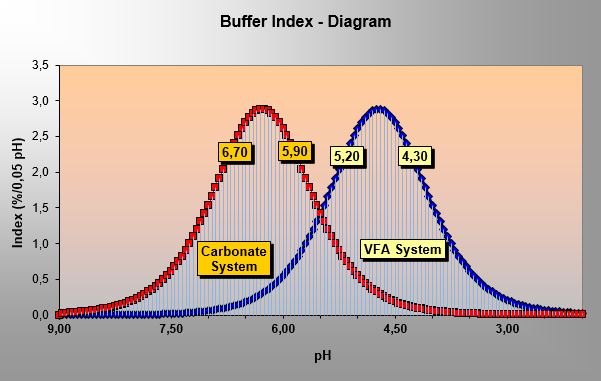
Volatile Fatty Acids / Alkalinity – Field Test
applied for early detection of biological process failure or overload in anaerobic reactors / digesters.
- Equipment: pH meter/probe, burette, magnetic stirrer, digital scale
- Procedure: no special training required – test is very user-friendly
- Calculations: data processing by PC using MS-Excel software
- Data recording: MS-Excel spread sheet
- Time requirement: approx. 10 minutes
Developed and distributed by IB-MR.

Equipment set-up
Advantages
- no sample dispatching
- no long waiting periods for results
- ‚plant failure’ due to overload can be ruled out if tests are carried out at regular intervals
- improved accuracy compared to other titration methods
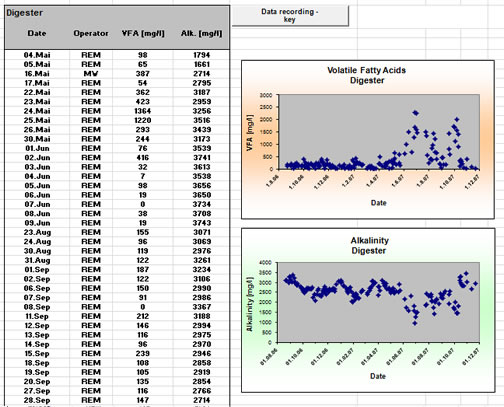
Data recording with MS-Excel
Acetic Acid Solutions
Solutions of acetic acid and sodium bicarbonate were made up to give acetic acid concentrations from 100 to 1000 mg/l and analyzed with the Five pH Point Titration. The results are plotted in Fig 1 (each point represents the average of five measurements).
Figure 1 illustrates the ability of the Five pH Point Titration to accurately measure acetic acid in made up solutions. For all test results the error remained within 10 % of the expected value.
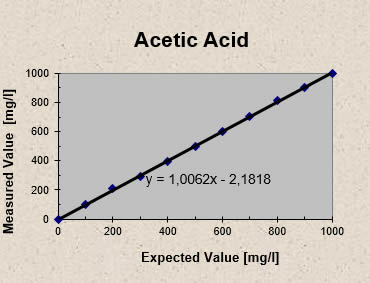
Fig 1 - Five pH Point Titration: Acetic acid recovery from made up solutions (acetic acid plus sodium bicarbonate)
Brewery Wastewater
Brewery wastewater samples augmented with known concentrations of acetic acid were analyzed with the Five pH Point Titration. The results plotted in Fig 2 show that for all samples the measured values did not deviate by more than 8 % from the expected values. Each point in Fig 2 represents the average of five titration results.
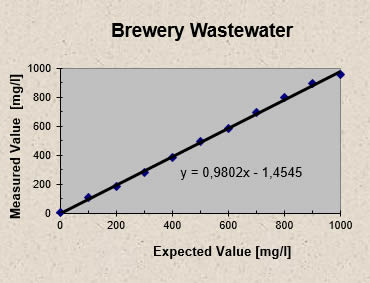
Fig 2 - Five pH Point Titration: Acetic acid recovery from brewery wastewater augmented with known concentrations of acetic acid
Conventional versus Five pH Point Titration
Results from a conventional titration method involving acid titration to pH 3, sample boiling and caustic soda back titration to pH 6.5 are compared to the results obtained by the Five pH Point Titration. Samples from a pulp mill anaerobic wastewater treatment plant were titrated using both methods and the results for acetic acid concentrations are plotted in Figure 3 - the conventional titration systematically overpredicts the volatile fatty acid concentration relative to the Five pH Point Titration. On average the conventional titration results exceed those of the Five pH Point Titration by 49 mg/l. Alkalinity results show a reverse trend: on average alkalinity results from the Five pH Point Titration exceed those of the conventional titration method by 23 mg/l, as illustrated in Figure 4.
For the purpose of plant monitoring the “error” is of little consequence. In this case, the Five pH Point Titration was selected as the preferred option, mainly, because of it is more user-friendly.
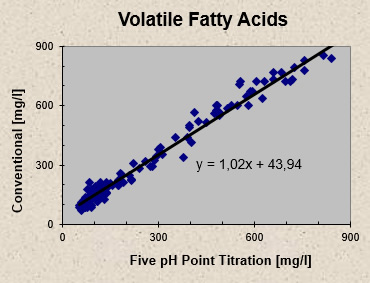
Fig 3 - Volatile Fatty Acids: Comparison between conventional and Five pH Point Titration
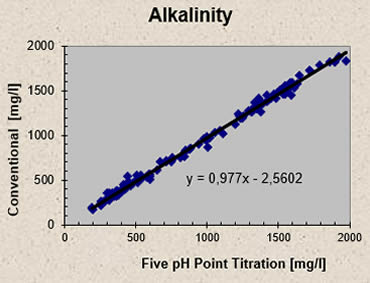
Fig 4 - Alkalinity: Comparison between conventional and Five pH Point Titration
Five pH Point Titration – Precision
Seventeen identical samples containing 300 mg acetic acid (HAc) and 1275 mg alkalinity (Alk) per liter were prepared and titrated using the Five pH Point Titration method. The averaged results were:
- 300 mgHAc/l (12 mg/l standard deviation)
- 1255 mgAlk/l (10 mg/l standard deviation)
Approximately 150 companies use the VFA/Alk Test (Five pH point titration) to monitor their anaerobic treatment plants.
The Five pH point titration method is recommended by Prof. Speece (Vanderbilt University, Nashville, US) in his book “Anaerobic Biotechnology for Industrial Wastewater”.
Theory and results were published in Water SA (South Africa), Water Science Technology (International Water Quality) and GWF Wasser-Abwasser.